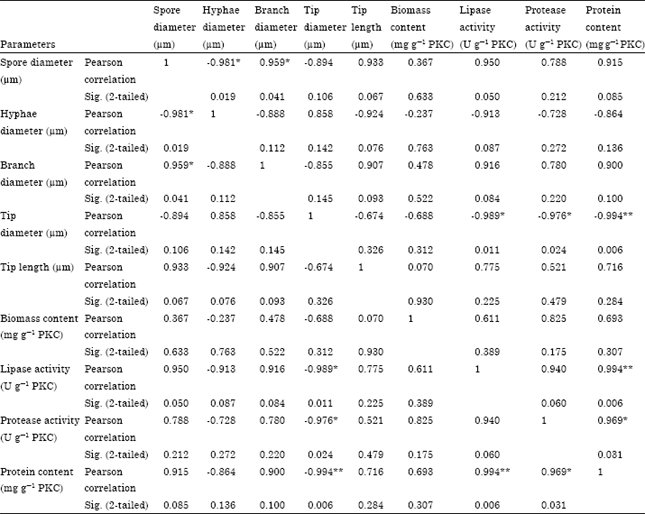
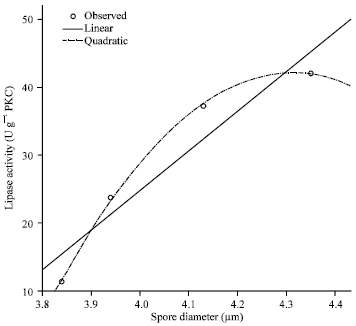
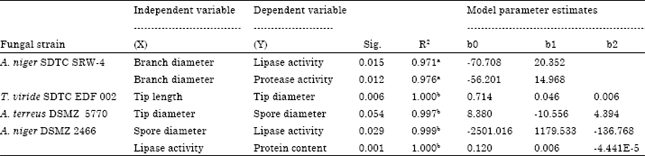
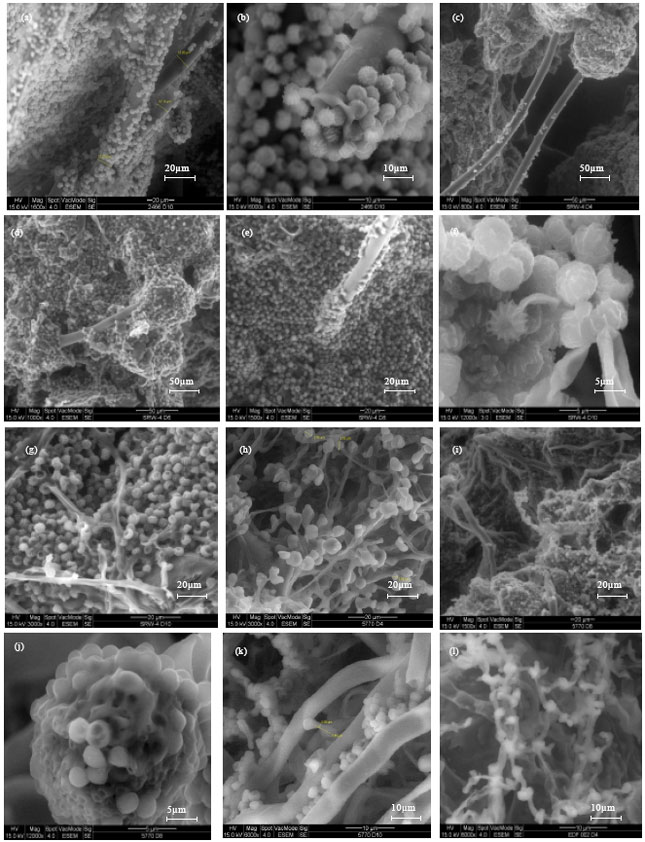
Research Article
Relationship between Fungal Growth Morphologies and Ability to Secrete Lipase in Solid State Fermentation
Sime Darby Technology Centre, UPM-MTDC III, Universiti Putra Malaysia, Serdang, Selangor, Malaysia
M.S. Umikalsom
Department of Bioprocess Technology, Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, Serdang, Selangor, Malaysia
T.C. Ling
Department of Food Process Engineering, Faculty of Engineering, Universiti Putra Malaysia, Serdang, Selangor, Malaysia
A.B. Ariff
Department of Bioprocess Technology, Faculty of Biotechnology and Biomolecular Sciences, Universiti Putra Malaysia, Serdang, Selangor, Malaysia