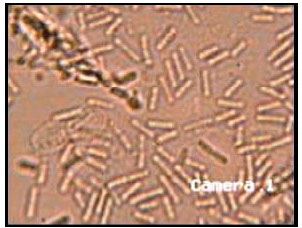
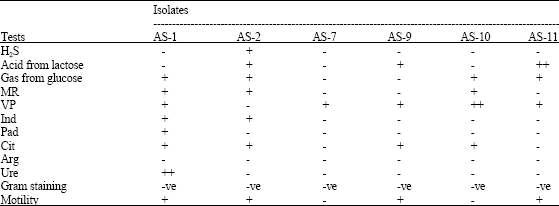
Research Article
Isolation, Optimization and Production of L-asparaginase from Coliform Bacteria
Atmiya Pharmacy College, Ankodia, Vadodara, Gujarat, India
R.V. Karadi
Department of Pharmaceutical Biotechnology, K.L.E.S�s College of Pharmacy, Belgaum-10, Karnataka, India
P.P. Parekh
JSMP�s Jaywantrao Sawant College of Pharmacy and Research, Hadapsar, Pune, Maharashtra, India