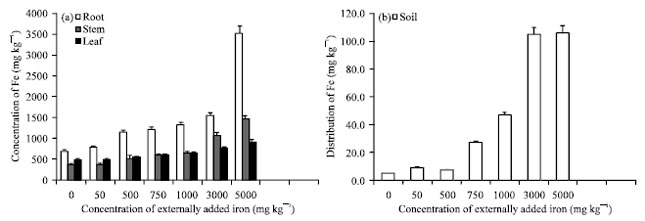
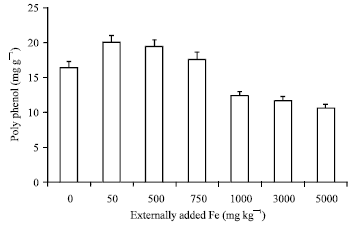
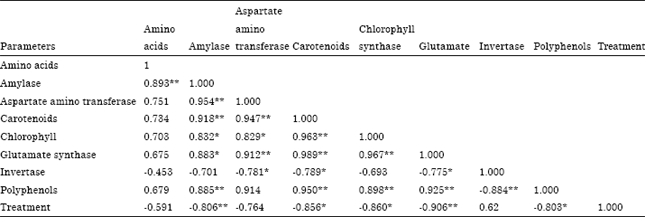
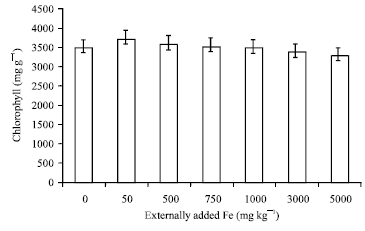
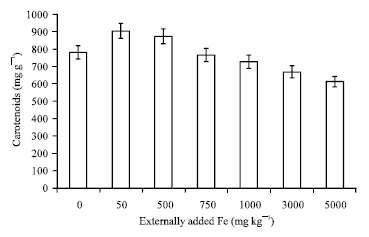
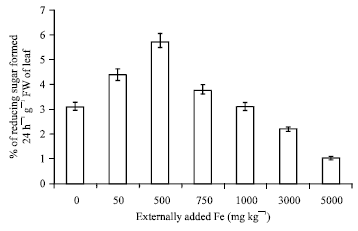
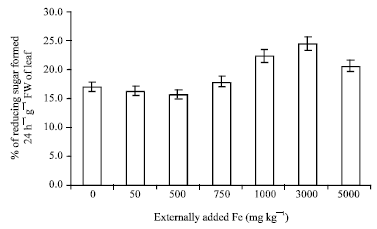
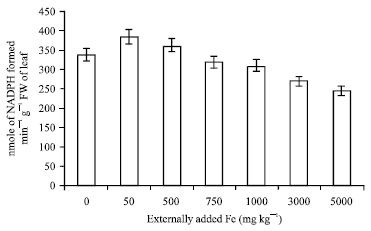
Research Article
Impact of Iron Toxicity on Certain Enzymes and Biochemical Parameters of Tea
K.S.R. College of Engineering, K.S.R. Kalvi Nagar, Tiruchengode-637 215, Tamil Nadu, India
Selvaraj Venkatesan
Measurements Laboratory, Gujarat Tea Processors and Packers Ltd., Vipul State, Khokhra, Ahmadabad-380008, Gujarat, India