Research Article
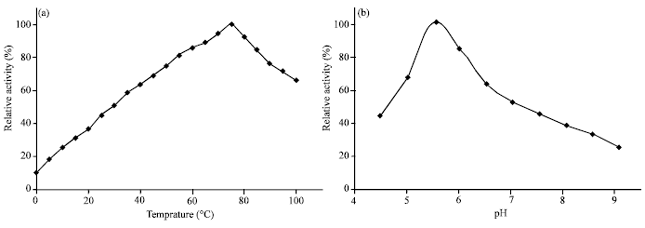
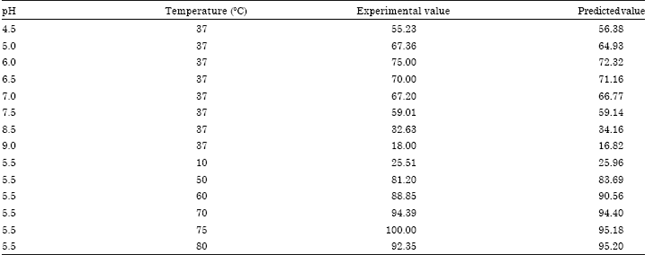
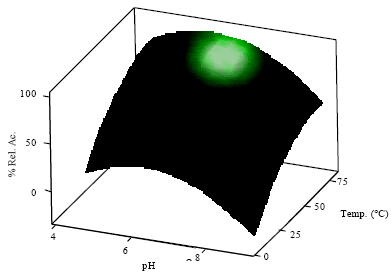
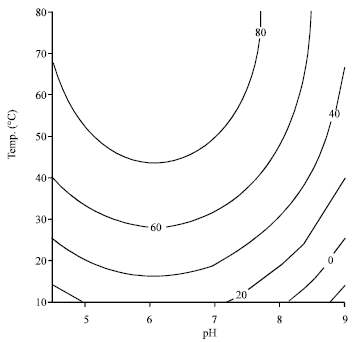
Application of Response Surface Methodology for the Determination of Optimum Reaction Conditions (Temperature and pH) for Starch Hydrolysis by α-Amylase
Department of Biochemistry, Institute of Technology, Banaras Hindu University, Varanasi-221005, U.P., India
Om Prakash
Department of Biochemistry, Institute of Technology, Banaras Hindu University, Varanasi-221005, U.P., India
Mahe Talat
Department of Physics, Nano-Sciences and Technology Unit, Faculty of Sciences, Institute of Technology, Banaras Hindu University, Varanasi-221005, U.P., India
S.H. Hasan
Department of Applied Chemistry, Institute of Technology, Banaras Hindu University, Varanasi-221005, U.P., India
Rajesh Kumar Pandey
Department of Biochemistry, Institute of Technology, Banaras Hindu University, Varanasi-221005, U.P., India