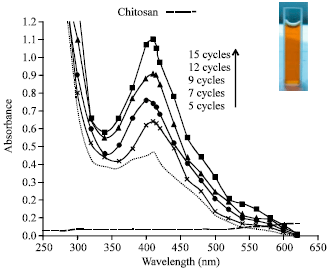
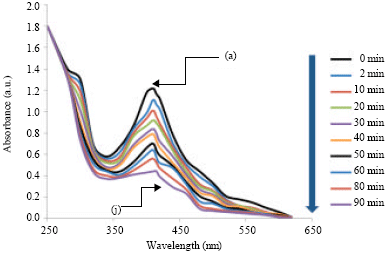
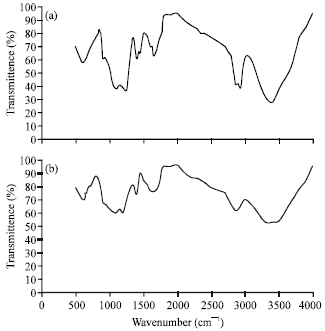
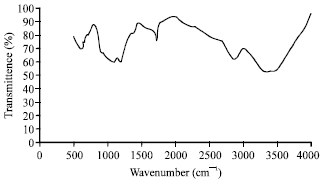
Research Article
Chitosan-silver Nanoparticles Composite as Point-of-use Drinking Water Filtration System for Household to Remove Pesticides in Water
Chemistry Unit, Department of Engineering Sciences, College of Engineering, Universiti Tenaga Nasional, Km 7, Jalan Kajang-Puchong, 43009 Serdang, Selangor, Malaysia
C.Y. Nian
Chemistry Unit, Department of Engineering Sciences, College of Engineering, Universiti Tenaga Nasional, Km 7, Jalan Kajang-Puchong, 43009 Serdang, Selangor, Malaysia
L.W. Zhan
Chemistry Unit, Department of Engineering Sciences, College of Engineering, Universiti Tenaga Nasional, Km 7, Jalan Kajang-Puchong, 43009 Serdang, Selangor, Malaysia
K.X. Ning
Chemistry Unit, Department of Engineering Sciences, College of Engineering, Universiti Tenaga Nasional, Km 7, Jalan Kajang-Puchong, 43009 Serdang, Selangor, Malaysia