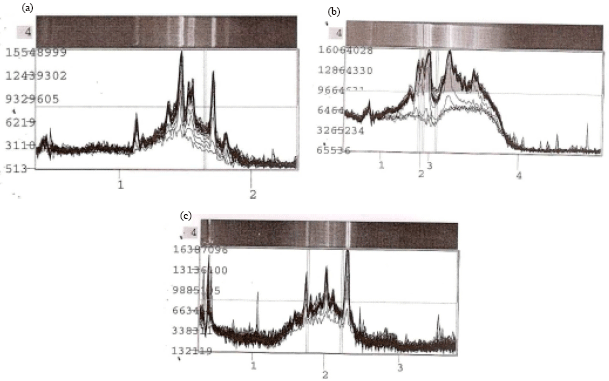
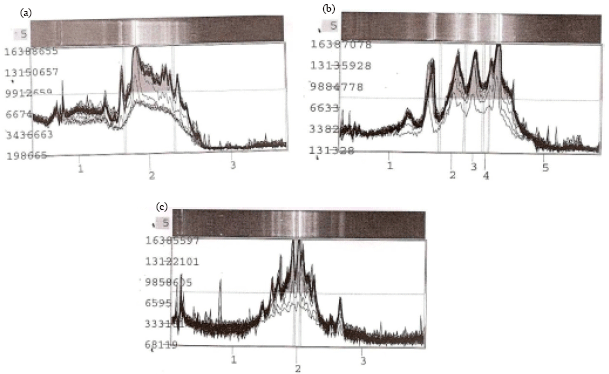
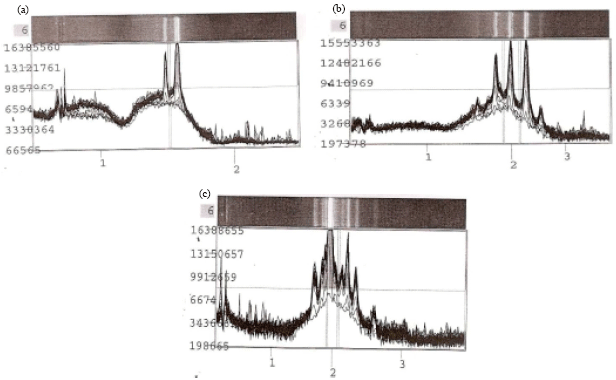
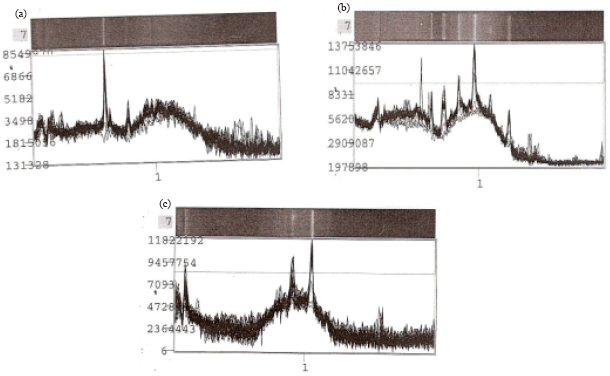
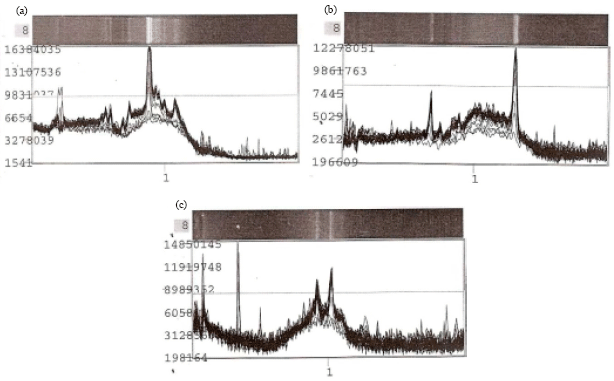
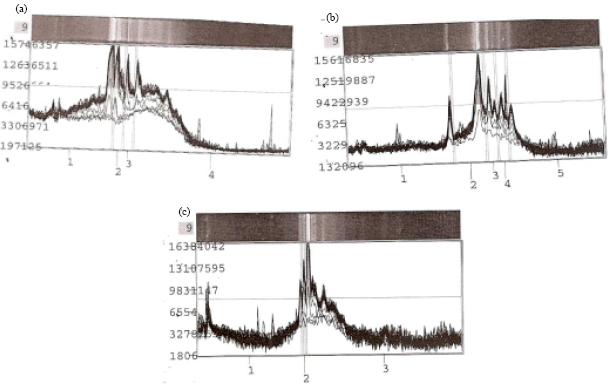
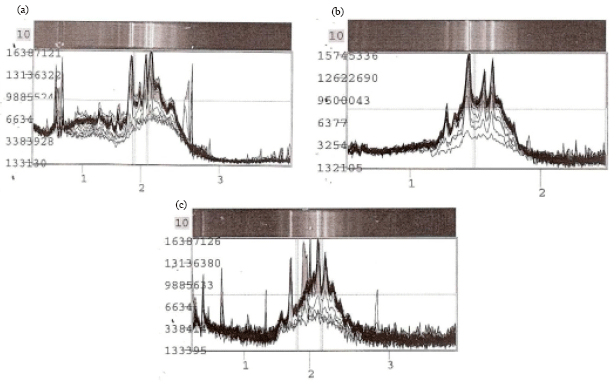
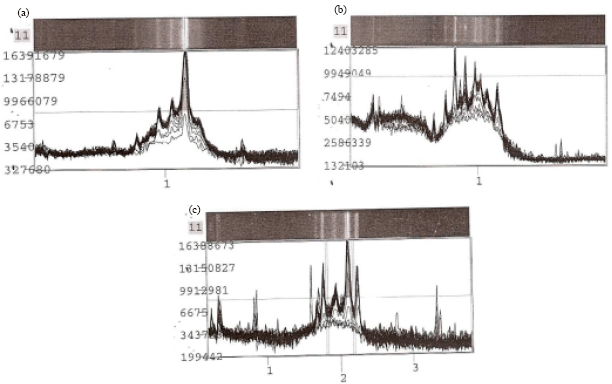
Research Article
A Phytochemical Profile of Albizia lebbeck (L.) Benth. Cultivated in Egypt
Department of Pharmacognosy, Faculty of Pharmacy, Cairo University, Cairo, Egypt
K. El-Fouly
National Organization for Drug Control and Research, Cairo, Egypt
N.M. Sokkar
Department of Pharmacognosy, Faculty of Pharmacy, Cairo University, Cairo, Egypt
Z. Talaat
National Organization for Drug Control and Research, Cairo, Egypt