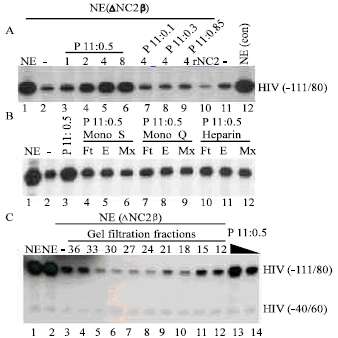
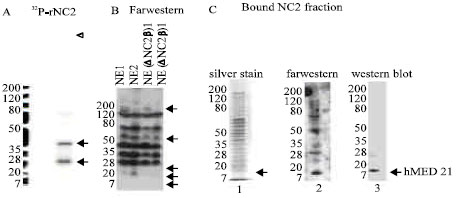
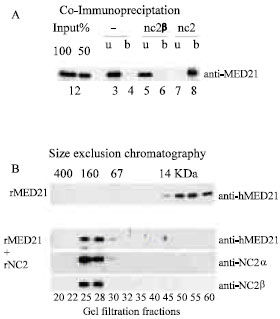
Research Article
Direct Interaction Between NC2 and a Subunit of the SRBs/mediator Co–activator
Unidad de Bioquirnica y Biologia Molecular de Plantas, Centro de Investigaci6n Cientifica de Yucatan, Calle 43 No. 130, CoL Chubwmi de Hidalgo, CP 97200, Merida, Yucatim, Mexico
Diana Guillen
Unidad de Bioquirnica y Biologia Molecular de Plantas, Centro de Investigaci6n Cientifica de Yucatan, Calle 43 No. 130, CoL Chubwmi de Hidalgo, CP 97200, Merida, Yucatim, Mexico
Luis Carlos Rodriguez
Unidad de Biotecnologia, Centro de Investigacion Cientifica de Yucatan, Calle 43 No. 130, CoL Chubwmi de Hidalgo, CP 97200, Merida, Yucatim, Mexico
Nadine Mobious
Unidad de Bioquirnica y Biologia Molecular de Plantas, Centro de Investigaci6n Cientifica de Yucatan, Calle 43 No. 130, CoL Chubwmi de Hidalgo, CP 97200, Merida, Yucatim, Mexico
Enrique Castano
Unidad de Bioquirnica y Biologia Molecular de Plantas, Centro de Investigaci6n Cientifica de Yucatan, Calle 43 No. 130, CoL Chubwmi de Hidalgo, CP 97200, Merida, Yucatim, Mexico