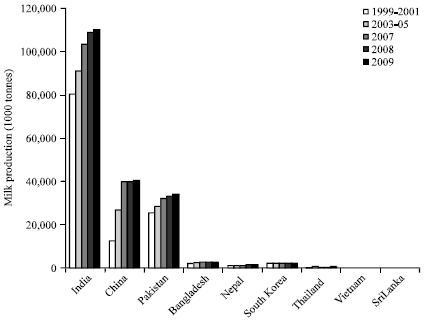
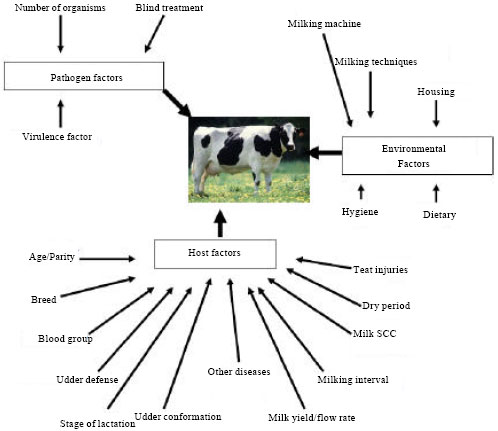
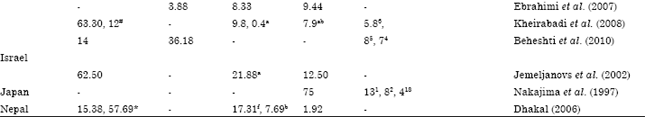
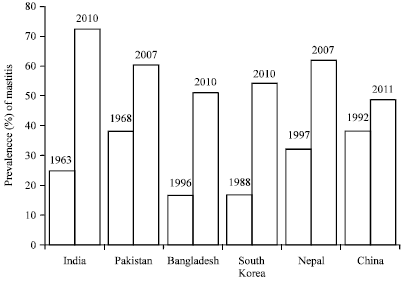
Review Article
Bovine Mastitis: An Asian Perspective
Department of Animal Biotechnology, College of Applied Sciences, Jeju National University, Jeju, South Korea
Gyu Jin Rho
College of Veterinary Medicine, Gyeongsang National University, Chinju, South Korea
Yeong Ho Hong
College of Veterinary Medicine, Gyeongsang National University, Chinju, South Korea
Tae Young Kang
College of Veterinary Medicine, Jeju National University, Jeju, South Korea
Hak Kyo Lee
Genomic Informatics Center, Hankyong National University, Ansung, South Korea
Tai-Young Hur
National Institute of Animal Science, Rural Development Administration, Cheonan, South Korea
Dong Kee Jeong
Department of Animal Biotechnology, College of Applied Sciences, Jeju National University, Jeju, South Korea