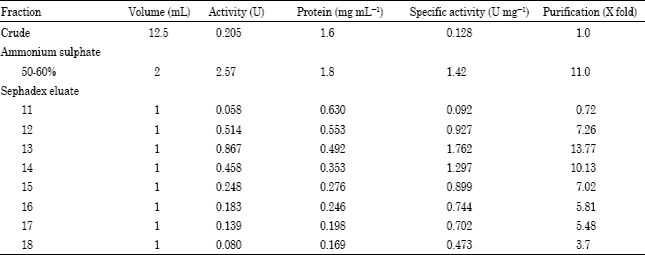
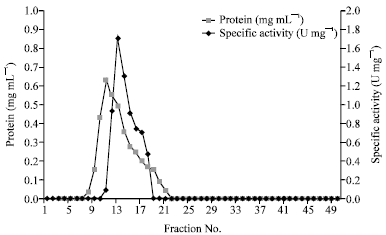
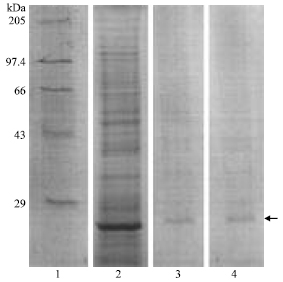
Research Article
Purification and Characterization of Plasmodium berghei Thioredoxin Reductase
Laboratory of Parasitology and Immunology, Department of Biosciences, Himachal Pradesh University, 171005, Shimla, India
H.S. Banyal
Laboratory of Parasitology and Immunology, Department of Biosciences, Himachal Pradesh University, 171005, Shimla, India