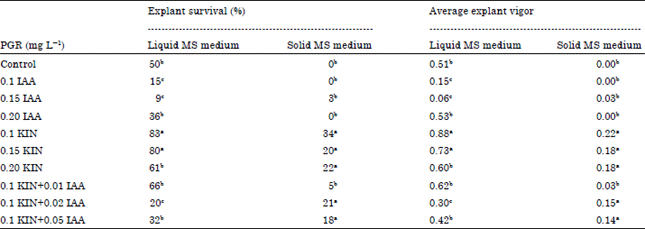
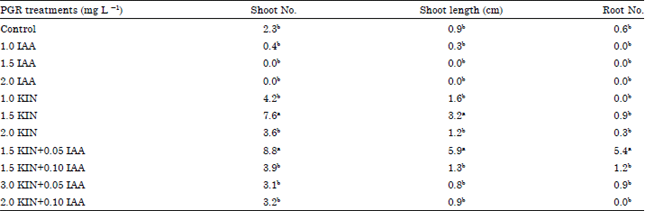
Research Article
Effects of Plant Growth Regulators on In vitro Regeneration of Malaysian Indica rice (Oryza sativa L.) cv. MR219 by Shoot Apical Meristem
Department of Biology, Faculty of Science, University Putra Malaysia, 43400 Serdang, Selangor, Malaysia
Rosimah Nulit
Department of Biology, Faculty of Science, University Putra Malaysia, 43400 Serdang, Selangor, Malaysia
Faridah Qamaruz Zaman
Department of Biology, Faculty of Science, University Putra Malaysia, 43400 Serdang, Selangor, Malaysia