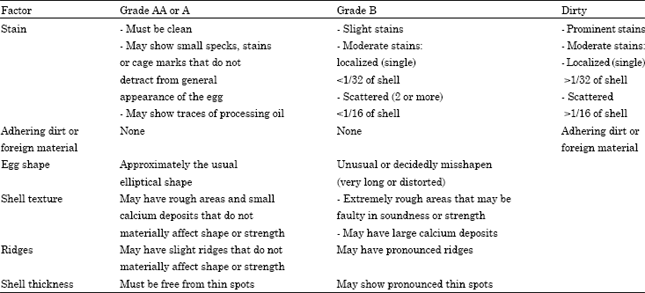
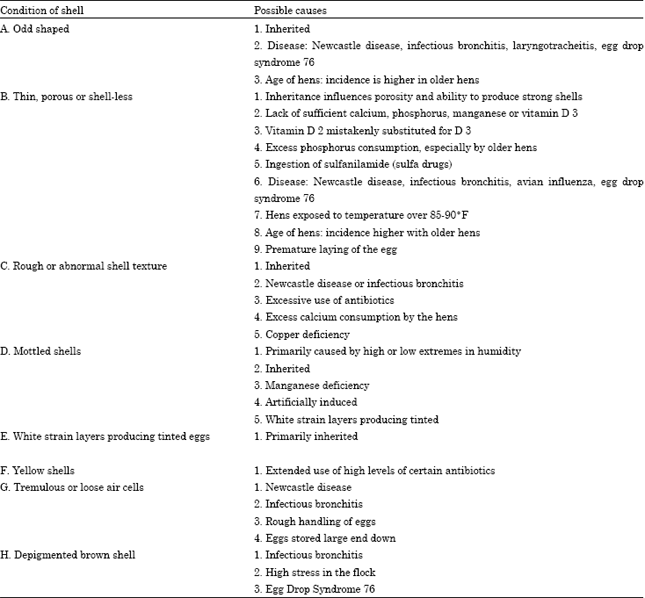
Review Article
Egg Quality Defects in Poultry Management and Food Safety
Department of Animal Science and Technology, Federal University of Technology, P.M.B 1526, Imo State Nigeria
I.C. Okoli
Department of Animal Science and Technology, Federal University of Technology, P.M.B 1526, Imo State Nigeria
N.J. Okeudo
Department of Animal Science and Technology, Federal University of Technology, P.M.B 1526, Imo State Nigeria
A.B.I. Udedibie
Department of Animal Science and Technology, Federal University of Technology, P.M.B 1526, Imo State Nigeria
I.P. Ogbuewu
Department of Animal Science and Technology, Federal University of Technology, P.M.B 1526, Imo State Nigeria
N.O. Aladi
Department of Animal Science and Technology, Federal University of Technology, P.M.B 1526, Imo State Nigeria
O.O.M. Iheshiulor
Department of Animal Science and Technology, Federal University of Technology, P.M.B 1526, Imo State Nigeria
A.A. Omede
Department of Animal Science and Technology, Federal University of Technology, P.M.B 1526, Imo State Nigeria