ABSTRACT
The need to prevent corrosion, toxicity and to enhance the aesthetic value of steel components in automotive, construction, electrical, appliances, recreational and materials handling industries and in our daily lives, has led to an enlarged interest in the field of electrodeposition of zinc on steel substrates. The toxicity and stringent regulation against water pollution and hence costly effluent disposal of cyanide based baths has led to the increased interest and accelerated growth of acid zinc based baths in the past few years. This study reviews the recent progress and development, bath solution and composition, effect and type of addition agents and practical aspects of acid based solutions. The corrosion resistance of the electrodeposited zinc coatings is also examined. The effectiveness of the cassava and sugar cane juices separately and the synergistic effect of the two juices combined is experimentally reported.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajaps.2012.314.326
URL: https://scialert.net/abstract/?doi=ajaps.2012.314.326
INTRODUCTION
Electrodeposition of zinc from acid solutions (chloride and sulphate baths) now constitutes about 45-50% of all zinc baths, particularly in the developed nations. Zinc is very electronegative and it provides sacrificial protection for steel substrates. It is very economical to deposit and worldwide supplies are high (Loto et al., 1991). Zinc is non-toxic and it is safe in contact with food. Other employed depositing solutions, apart from acid solutions, are those based on cyanide baths and to a lesser extent fluoborate, alkaline zincate and pyrophosphate (Fraunhofer, 1976).
Cyanide zinc baths are the most widely used, have high throwing power, which is one of the important factors in zinc plating. However, due to their toxicity and the stringent regulations against water pollution and costly effluent disposal, non-cyanide and low-cyanide baths have been investigated and used for commercial plating (Schlesinger and Paunovic, 2000; Vagramyan et al., 1979). The trend of replacing the cyanide with non-cyanide baths has been steady for the past fifteen years, however, it has accelerated greatly in the last six years. Some recent surveys have shown that the non-cyanide zinc baths now out-number their cyanide counterparts by more than two to one. Improvements in alkaline zincate, acid sulphate and acid chloride baths have been reported in recent years (Geduld, 1998; Bapu et al., 1998; Saubestre and Hadju, 1974; Pushpavanam et al., 1981). A typical constituent concentration of cyanide baths (Fraunhofer, 1976; Lowenheim, 1963; Gabe, 1978) consists of zinc (as metal), 25-60 g L-1, sodium cyanide 10-150 g L-1, sodium hydroxide 25-140 g L-1 and operating temperature of 15-60°C.
Non-cyanide zinc plating solutions can be divided into two types, mildly acid solutions (using chloride or sulphate anions) and alkaline-zincate solutions (Darken, 1979). The mild baths generally consist of zinc chloride dissolved in a solution of excess ammonium chloride (more recently potassium chloride processes, which are far less corrosive have been marketed and ammonia free formulation is now the most popular in production (Marcos and Bertazzoli, 1986). Zincate baths consist solely of a small concentration of zinc metal dissolved in approximately 100 g L-1 sodium hydroxide solution (Darken, 1979). Chloride zinc solution does not only eliminate cyanide in plating, it also gives improved bath efficiency and exceptional brightness. Acid zinc baths are used where it is desirable to have a high plating rate and low cost.
Chloride zinc plating offers considerable advantages over cyanide based systems, although, it is not without its share of routine operating problems (Schneider, 1987). Use of the acid zinc sulphate process is increasing due to its relatively low cost, safety features and pollution control characteristics, but poor throwing power and insufficient brightness from acid sulphate bath are disadvantages (Vagramyan et al., 1979).
This study examines the recent progress made in understanding and improving bright deposition from acid chloride and sulphate solutions. It covers areas such as bath solution composition, effect and types of addition agents/brightness, practical aspects of acid based solutions-cathode efficiency, throwing power (deposits from acid solutions and operating problems) and corrosion resistance of the electrodeposited zinc coatings.
The unique effectiveness of addition agents obtained locally from the juices of cassava tuber and sugar cane is also experimentally presented.
THE ACID ZINC PLATING SOLUTIONS
Acid chloride zinc baths currently in use are principally of two types (Geduld, 1982): Those based on ammonium chloride and those based on potassium chloride. The ammonium-based baths were the first to be developed. They can be operated at higher current densities than potassium baths.
Marcos and Bertazzoli (1986) has described three types of contemporary chloride baths. These are listed in Table 1. The ammonia-free (potassium) referred to as Type I bath has been described as the most popular in production.
The use of low (or mini) ammonia bath comes next. The ammonium chloride replaces boric acid at a concentration of about 30 g L-1. these baths offer the advantage of improved solubility of ammonium vs. boric acid, a wider higher-current-density plating range at a low metallic zinc levels and high temperature capabilities (Marcos and Bertazzoli, 1986). The full-ammonia bath which author has described as Type 3, finds little use and most have been converted to the low-ammonia or ammonia-free variety.
In the low ammonia and ammonia free baths, there is limited use of sodium chloride in place of potassium chloride. This is because the sodium chloride electrolyte has reduced cathode efficiency and is more corrosive when compared with potassium chloride. Also, the solubility of proprietary addition agents is reduced in the sodium chloride solution.
The concentration of the constituents for chloride zinc baths also varies (Preiksaite and Sarmaitis, 1981) as indicated in Table 2. All bright acid chloride processes are proprietary and some degree of incompatibility may be encountered between them.
Zinc sulphate is used as a source of metal, ammonium chloride increases the conductivity, sodium acetate acts as a buffer and glucose acts as an addition agent.
| Table 1: | Chloride bath types |
 | |
| Table 2: | Concentration of constituents of chloride zinc baths |
 | |
| *Proprietary additive for zinc plating with the trade name Limeda, Institute of Chemistry and Chemical Techn., Academy of Sciences of Lithuania SSR, Vilnius, USSR (Preiksaite and Sarmaitis, 1981) | |
| Table 3: | Varieties of acid zinc sulphate |
 | |
Table 3 illustrates the different varieties of acid zinc sulphate as reported in different relevant literature. However, the coating is not bright, but mat and hence different additives are required to improve the throwing power of acid zinc sulphate baths.
A typical old formula for acid zinc sulphate bath (Blum and Hogaboom, 1930) is indicated in Table 4.
| Table 4: | A typical old formula for acid zinc sulphate bath |
 | |
ADDITION AGENTS-BRIGHTENERS
Addition agents for acid zinc chloride bath have been mainly proprietary. In his review, Marcos and Bertazzoli (1986) described most secondary brighteners as consisting of aldehydes or unsaturated ketones. These active ingredients were usually made soluble with alcohol or other solvents. Bisulphite addition products which introduced sulphur compounds to the bath were employed in an early attempt to formulate water-soluble brighteners. These compounds were often detrimental especially in rack plating. Microemulsion technology, today, produces completely water soluble brighteners without unnecessary side products. It is also known that solublization by this method increases brightener mileage with the same level of active ingredients. In the bath, the brightener is more quickly activated and as it was with the older solvent systems, automatic feed hardware is not attacked (Marcos and Bertazzoli, 1986). A detailed discussion of how aldehydes and other brighteners react at the zinc cathode during deposition was given (Defonte, 1978).
The development of different organic addition agents has enabled bright zinc electrodeposits to be produced from acid zinc sulphate baths. In a study (Venkatesha et al., 1987) to find a new brightener for the acid sulphate bath, an addition of furfural gave satisfactory mirror bright zinc deposits. Deposit from standard solution with 1 mL L-1 of furfural, i.e., the bath that contained both dextrin have a fine-grained structure and bright deposits were obtained over a wide range of current density.
Glycine and thiourea have also been used as brighteners in acid zinc sulphate baths (Venkatesha et al., 1987). The concentrations of brighteners were determined during electrolysis by colorimetric (Hiremath and Mayanna, 1984) and volumetric (Uma and Mayanna, 1980) procedures using tri-beta hydrindene hydrate (ninhydrin) reagents and chloramine-B, respectively. The uniformity of zinc deposits with a fine grain size was improved by carrying out Hull Cell experiments using the standard bath (Table 5), plus various organic addition agents such as glucose (0.5 to 5.0 g L-1), starch (0.5 to 5.0 g L-1), gelatin (0.1 to 2.0 g L-1) and dextrin (0.5 to 5.0 g L-1). A white, uniform, fine-grained zinc deposit was produced over a wide current density range using 3 g L-1 dextrin. The other addition agents produced grey deposits.
New zinc brighteners for the acid sulphate bath was found by using several oxygen-, nitrogen-and sulphur-containing organic compounds in the bath and a Hull Cell zinc pattern was examined in each case. Glycine in combination with thiourea was found to be an effective brightener over a fairly broad range of current densities. The optimum bath composition and operating conditions used are indicated in Table 6.
The synergistic effect of additives on bright nanocrystalline zinc electrodeposition was investigated (Nayana et al., 2011). The authors studied the influence of additives like cetyltrimethylammonium bromide (CTAB) and Ethyl Vanillin (EV) on zinc electrodeposition from acid sulphate bath by scanning electron microscopy, X-ray diffraction and voltammetric techniques. Their result showed the existence of interaction between CTAB and EV.
| Table 5: | Standard bath composition and operating* conditions (Venkatesha et al., 1987) |
 | |
| *Plated in hull cell for 5 min at 2A using air-agitated bath at 30°C and pH 2.5, a mild steel cathode and an anode of 99.9% zinc sheet | |
| Table 6: | Optimum bath composition and operating condition (Venkatesha et al., 1987) |
 | |
 | |
| Fig. 1(a-d): | SEM images of deposit obtained from bath (a) I, (b) II, (c) III and (d) IV (Nayana et al., 2011) |
| Table 7: | Zinc plating bath (Nayana et al., 2011) |
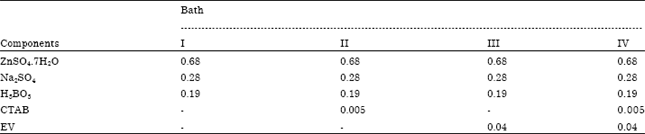 | |
They exhibited synergistic effect to produce bright nanocrystalline zinc coating on steel surface. The nano-sized bright crystalline zinc deposit was obtained from sulfate electrolyte containing both ethyl vanillin and cetyltrimethylammonium bromide. The deposit transformed from dull appearance (without additives) to smooth fully bright appearance in the presence of both additives (Fig. 1). The bath composition used for zinc electrodeposition is given in Table 7.
PRACTICAL ASPECTS OF ACID ZINC BASED SOLUTIONS
Cathode efficiency: The high cathode current efficiencies exhibited by chloride zinc baths are one of the most important properties of these baths (Geduld, 1982). The average cathode current efficiency for these baths is approximately 95 to 98% over the entire range of operable current densities. There is no any other zinc plating system that approaches this extremely high efficiency at higher current densities. The high cathode efficiency combined with the quick brightening action provide for faster plating and increased productivity (Marcos and Bertazzoli, 1986). The high efficiency can lead to productivity increases of 15 to 50% over cyanide baths.
As shown in Fig. 2, the alkaline non-cyanide and cyanide zinc baths give respectable cathode efficiencies at low current densities. As the current density increases, a dramatic drop off is indicated. On the other hand, chloride zinc maintains very high cathode efficiency across the entire current density range, so the high-current-density areas of parts are subject to very high deposition rates. This results into much more mental being plated in the high-than in the low-current density regions or, simply put, the bath displays poor throwing power (Marcos and Bertazzoli, 1986).
Throwing power: The throwing power on all the different types of zinc electroplating solutions have been measured (Paatsch and Hogaboom, 1980). It has been noted that throwing power is dependent on the both primary and secondary current distributions, the former emanating from work piece shape, arrangement of electrodes and dimensions of the bath. Todt has quoted the following throwing power values determined in the Haring-Blum Cell:
 |
The above clearly shows the inferior power of acid zinc solutions. The work undertaken by Blum and Paatsch on the depth of penetration of a zinc deposit into a steel tube, with the open end directly facing the anode, has been described (Darken, 1979). Throwing power under a standard condition was of the following order:
High cyanide>Low>Acid>Alkaline cyanide free |
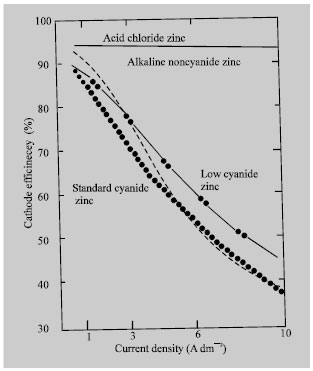 | |
| Fig. 2: | Comparison of cathode current efficiencies of bright zinc plating electrolytes (Geduld, 1982) |
An alternative method of comparing deposit distribution from various zinc plating solutions by determining the thickness deposited during Hull Cell plating tests at various distances from the high current density edge was indicated (Darken, 1979). It was indicated from the results obtained that acid zinc solution gave a deposit with least favourable thickness distribution.
CORROSION RESISTANCE
Several investigations (Preiksaite and Sarmaitis, 1981; Pushpavanam, 1986; Budman, 1995; Beltowska-Lehman et al., 2002; Adaniya et al., 1980, 1981; Leidheiser and Suzuki, 1981) have been carried out to improve the corrosion resistance of acid sulphate bath electrodeposited zinc. Corrosion resistance, twice as high as pure zinc, was obtained in the deposits by the additions of cobalt and chromium. Extensive investigations into the corrosion resistance of chromate and uncoated zinc electrodeposits were carried out (Preiksaite and Sarmaitis, 1981). Salt spray tests for 9 μm coated samples showed that the service life decreased in the following order: bright cyanide>neutral matt>matt cyanide>bright-zincate>bright weak acid, ammonium chloride>bright, weak acid, ammonium free. It has been shown that zinc coatings deposited from baths of different composition are not alike; they differ in porosity, structure and other characteristics (Todt, 1998). These in turn, should affect the corrosion resistance of the coatings.
The effect of MoS2 on the deposition properties, morphology, crystallographic orientation and corrosion behaviour of the electrodeposited Zn-MoS2 nanocomposite coatings on mild steel from zinc sulphate-chloride bath containing uniformly dispersed MoS2 nanoparticles were studied (Kanagalarasa and Venkatesha, 2011) (Fig. 3).
 | |
| Fig. 3(a-b): | Image of (a) Zinc and (b) Zn-MoS2 (1 g L-1) coating (Kanagalarasa and Venkatesha, 2011) |
It was shown that the addition of MoS2 to the electrolyte significantly changed the microstructure and crystallographic orientation of the zinc deposits and enhanced the corrosion resistance of the coatings. The morphological and electrochemical properties of the zinc coatings were observed to be significantly affected by the incorporation of MoS2 particles into the zinc matrix.
Zemanova (2009) investigated the corrosion resistance of zinc coatings in an accelerated corrosion test in a condensation chamber. Zinc was electrodeposited from alkaline and acidic electrolytes using direct current DC or Pulse Current (PC). The zinc coating was subsequently protected against corrosion with a chrome (III) layer.
Morphology and structure of the coatings was investigated using Scanning Electron Microscopy (SEM), Energy Dispersive X-ray Spectroscopy (EDX) and X-ray diffraction analysis (XRD) before and after the corrosion test. Corrosion resistance of alkaline zinc coatings electrodeposited with DC and PC under test conditions was found to be comparable. The corrosion resistance of zinc coatings deposited from acidic electrolytes with PC was lower in comparison with corrosion resistance of zinc coatings deposited using DC. Excellent corrosion resistance of the tested coatings in a condensation chamber was achieved for samples PC-electrodeposited from both zinc acidic and alkaline electrolyte with the frequency ratio of current-on to current-off time equal to 1 at the electrodeposition time of 10 min. The samples were treated with trivalent chromium. The quality of PC-electrodeposited samples corrosion resistance was found to be comparable to that of samples obtained by the DC electrodeposition using commercial electrolytes. However, the PC electrodeposited coatings are not bright enough due to frequency modification, as it is typical for DC electrodeposition due to the presence of organic additives. The author was of the opinion that inhomogeneous character of the samples morphology, in the sense of sheet or leaf-like grains obtained using PC in the frequency range of 22-90 Hz, could be the reason for the corrosion attack to the white rust. It was observed, in general, that zinc coatings obtained by PC electrodeposition from alkaline electrolytes provide more corrosion-resistant coatings in comparison to those PC-electrodeposited from acidic electrolytes under the test conditions. The author’s results and observations were supported with SEM micrographs as presented in Fig. 4.
 | |
| Fig. 4(a-c): | SEM micrographs of electrodeposited zinc from acidic and alkaline electrolytes using (a) DC, (b) PC 50 Hz and (c) PC 22 Hz (Zemanova, 2009) |
DEVELOPMENT OF NEW ADDITION AGENTS
The effects of thiourea, dextrin and glycine additives, as organic addition agents, on the surface characteristics of zinc electrodeposition on mild steel in acid chloride solution at different pH levels, varying combinations of the addition agents, at varying and constant time, had been studied by the Scanning Electron Microscopy (SEM) examinations (Loto and Olefjord, 1992). The overall results show a good zinc electrodeposition on mild steel, though the obtained unique crystal structures were characteristic of the different pH level, varying time and different combinations of addition agents. The above mentioned addition agents have been previously used in the acid sulphate baths.
The `green’ addition agents-experimental: This last part of the paper reviews the results of a study of the performance of cassava and sugar cane juice extracts as addition agents in acid chloride solution. The detailed experimental study was reported in a previous publication (Loto, 1993).
Flat mild steel-SIS 141147, 0.1 cm thick, with a nominal composition of 0.038% C, 0.19% Mn and the rest Fe, was cut into several test specimens of 10.0 cm long and 1.0 cm wide. A portion of 1.0 cm in length was marked off at one end for the electrodeposition of zinc.
The test specimens were degreased ultrasonically for 5 min with an alkaline degreasing chemical, code named Henkel VR 63 62-1 and then removed from the solution rinsed in distilled water, immersed in methanol and air dried. The specimens were, in turns etched for 50 sec in 3 M HCl, rinsed in distilled water, immersed in methanol, air dried and stored in a desiccators for further experimental process. The acid chloride solution for the electrodeposition consists of ZnCl (71 g L-1), KCl (207 g L-1) and H3BO3 (35 g L-1). Extracted cassava and sugar cane juices, each (25-30 mL L-1 of acid chloride, solution) were used as the addition agents.
Electrodeposition of zinc on steel was performed by partially immersing the steel specimen and the zinc electrodes in the plating solution (20 mm deep) through the rectangular hole made on a perspex plastic cover for the 250 mL beaker used as the plating bath. The steel specimen was connected to the negative side of a DC supplier while the zinc electrodes were also connected with a wire. The plating solutions were put, in turns, into the beaker (bath) and their respective pH was obtained by adjusting the original solution with potassium hydroxide. Four different plating baths were used. These consist of:
| • | Plating from the acid solution without the addition agents |
| • | Plating from the solution with cane sugar juice only (in 25 mL L-1) as addition agent. |
| • | Plating from solution with cassava juice (in 30 mL L-1) alone as the addition agent and, |
| • | Plating from solution with cassava and sugar cane juices (30 mL each) as the addition agent |
The operating conditions were:
| pH of the solution | = | 4.8-5.2 |
| Temperature | = | 28-30°C |
| Current | = | 3.0 mA cm-1 |
| Plating time | = | 15 min |
A gentle stirring was used during the plating operations.
After each zinc electrodeposition, the plated specimen was taken out, rinsed in distilled water, immersed in methanol and quickly air-dried before the surface photograph was taken (Fig. 5). The unavailability of scanning electron microscope prevented the plated specimen surface from being characterized. The adhesion of the zinc coating to the steel substrate was tested by using a cellotape fastened to the surface and later pulled off and visually observed for any zinc stripping from the plated steel’s surface. The very acidic plating medium could easily destroy the unstable surface film of the metal specimen and rendered the steel’s surface bare to serve as a good zinc electrodeposition substrate.
Photomacrograph of the as-received unplated specimen is presented in Fig. 5a. The plated specimen without any of the addition agents-cassava and sugar cane juices. Fig. 5b, did not show bright plating; the plating was dull and though, some how effective as expected. In Fig. 5c, which shows the plated specimen with the cassava juice as addition agent in the plating medium is effective to some extent. Similar result was obtained for the addition of sugar cane juice (Fig. 5d). The effectiveness of these two additives was actually due to their chemistry.
Cassava juice is known (Coursey, 1983; Conn, 1983; Nastey, 1983) to contain about 93% of the cyanogenic glycoside linamarin-2 (β d-glucopyranosyloxy) isobutyronitrile with about 7% of the closely related lotaustralin-2 (β-d-glucopyranosyloxy) 2-methyl butyronitrile. During fermentation, these substances hydrolyse under the influence of the endogeneous enzyme linamarase to liberate hydrogen cyanide. Cyanide ions in the cyanide plating bath is known to give the brightest zinc plating.
Though making an effective contribution, the very low percentage of cyanide ion (CN-) in cassava juice, however, did not allow it to give the very bright plating characteristic associated with the cyanide bath. Sugar cane juice consists of sucrose, a non-reducing sugar. Sucrose has been known to be α-D-glucopyranosyl-β-D fructose (Finar, 1969). Sucrose can be hydrolysed by dilute acids such as in this acid zinc bath or by the enzyme invertase to an equimolecular mixture of D (-) fructose.
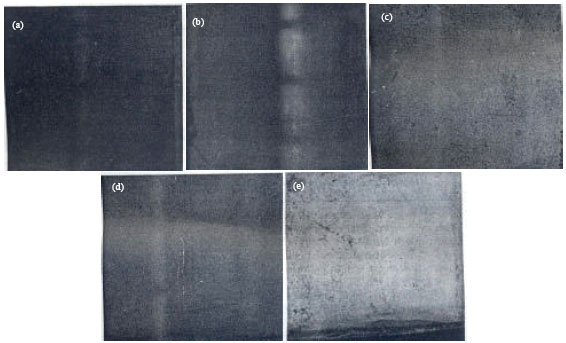 | |
| Fig. 5(a-e): | Photomacrograph of the unplated and plated surfaces of the mild steel specimen: (a) unplated mild steel specimen, (b) plated mild steel without addition agent, (c) plated mild steel with cassava juice as addition agent, (d) mild steel with sugar cane juice as addition agent and (e) plated mild steel with cassava and sugar cane juices as addition agent (Loto, 1993) |
The two monosaccharide molecules are linked through their reducing groups. Glucose has been previously used and is still being used as addition agent in zinc electroplating.
The synergistic effect of the combined use of cassava and sugar cane juices is apparent as presented in Fig. 5e. The chemistry of the cassava juice combined with that of the sugarcane juice would have given a chemical complex that became very effective in giving a good zinc electrodeposition on steel.
CONCLUSION
The continuing development of acid zinc plating baths based on zinc chloride has radically altered the technology of zinc plating which now constitutes about 50% of all zinc baths worldwide. It is the fastest growing baths throughout the world. A lot of development has been made in improving the brightness of the acid sulphate bath and also in improving the corrosion resistance of electrodeposits from acid baths. The inherent major advantages have contributed to the recent trend of growth in the acid baths. A good zinc electrodeposition on mild steel surface could be obtained in the acid zinc chloride solution using either the cassava juice or the sugar cane juice extract alone. However, the combination of the two gives a far better synergistic result.
REFERENCES
- Bapu, R.G., G. Devaraj and J. Ayyapparaj, 1998. Studies on non-cyanide alkaline zinc electrolytes. J. Solid State Electrochem., 3: 48-51.
Direct Link - Marcos, R.V.L. and R. Bertazzoli, 1986. Selection of a commercial anode oxide coating for electro-oxidation of cyanide. J. Braz. Chem. Soc., 13: 3-3.
Direct Link - Beltowska-Lehman, E., P. Ozga, Z. Swiatek and C. Lupi, 2002. Influence of structural factor on corrosion rate of functional Zn-Ni coatings. Cryst. Eng., 5: 335-345.
Direct Link - Hiremath, R.C., S.M. Mayanna, 1984. Kinetics and mechanism of oxidation of chloramphenicol by 1-chlorobenzotriazole in acetic medium. J. Chem. Sci., 117: 333-336.
Direct Link - Kanagalarasa, V. and T.V. Venkatesha, 2011. Studies on electrodeposition of Zn-MoS2 nanocomposite coatings on mild steel and its properties. J. Solid State Electrochem.
CrossRef - Leidheiser, H. and I. Suzuki, 1981. Cobalt and nickel cations as corrosion inhibitors for galvanised steel. J. Electrochem. Soc., 128: 242-249.
Direct Link - Venkatesha, T.V., J. Balachandra, S.M. Mayanna and R.P. Dambal, 1987. Effect of glycine and thiourea as brighteners in acid zinc sulfate bath. Plat. Surf. Finish., 74: 77-80.
Direct Link - Vagramyan, T., J.S.Li. Leach and J.R. Moon, 1979. The structures of zinc electrodeposits formed at low current densities. J. Mater. Sci., 14: 1170-1174.
CrossRefDirect Link - Zemanova, M., 2009. Corrosion resistance of zinc electrodeposited from acidic and alkaline electrolytes using pulse current. Chem. Papers, 63: 574-578.
CrossRefDirect Link









Michael Binder Reply
Highly informative paper.
I am particularly interested in low cost, green additives (such as sugar cane juice) to improve performance.
Michael Binder
Prof. C.A. Loto
I am indeed pleased and encouraged with the comment of Michael Binder. There are other series of articles based on experimental studies that will soon be out from some different international Journals.
satender Reply
dear sir,i am facing problem regarding acid zinc plating barrel type.zinc plates on both side are not working inspites of various efforts i/c current density and chemicals check up